Medorganics India is licensed by the AYUSH (India) and FSSAI to manufacture, pack and market various herbal products in different dosage forms (Prescription drugs, OTC, Herbals) respectively.
Raw material and packing materials Receipt.
RM & PM on receipt shall be dedusted & verified for receipt quantity, inwarded & Labelled.
Storage of raw materials and packing materials
The RM/PM shall be stored separately in the dedicated & Labelled Quarantine areas respectively.
Sampling, Testing and Release
Sampling of Raw Materials shall be carried out by QC personnel, in Sampling both.
Sampled RM/PM shall be Tested according to Pharmacopoeial/In-House Methods, on conformation to Quality Standards the RM/PM shall be Released with Approved Label. The Approved materials shall be Shifted to dedicated Approved Storage areas respectively.
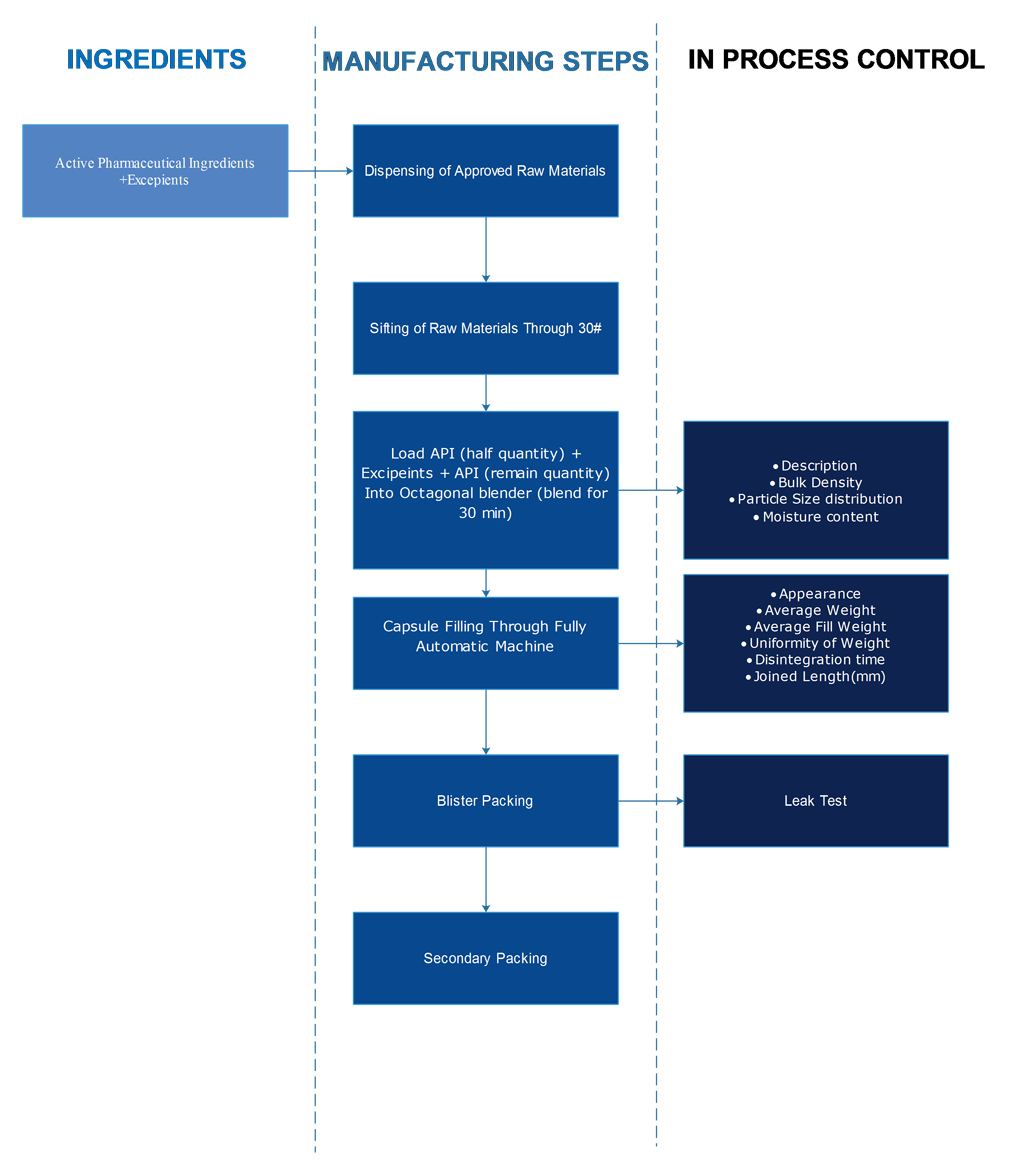
Dispensing
The Approved RM’s shall be dispensed under Dispensing Booth, in accordance to the Standard Quantities of the Batch Manufacturing Record and Passed on to the Granulation area Through the Pass box.
Manufacturing
Granulation
The Dispensed Materials shall be verified against Standard quantities.
The RM shall be Sifted through Sifter and Mixed for ingredients uniformity in Rapid Mix Granulator. Binder shall be added for the formation of Granules. Wet Granules shall be dried in Fluidised Bed Drier to reduce the Moisture content. Finally Milled and sifted for size uniformity.
Blending
The dried granules shall be mixed lubricants, disintegrants and glidants for uniform mixing dosage form in a blender.
Tablet Compression
The Blend shall be punched into desired shaped Tablets through the Compression machine, the tablets shall be dedusted to remove the adhered dust particles through deduster and passed through Metal detector to separate tablets containing metal particles, if any.
Coating
The Compressed Tablets shall be coated with approved food grade colourants by spraying coating solution in an autocoater machine, to provide protection from damage.
Capsule Filling
The blended materials / pellets shall be filled into hard cellulose / hard gelatine capsules through a fully automated capsule filling machine followed by automated polishing and sorting of good capsules.
Tablet Inspection
The defective, decolured, chipped or any kind damaged tablets shall be sorted out and rejected during Tablet inspection process. Only the Good tablets shall be proceeded to the next stage.
Blister packing
15 Tablets or 10 Tablets shall be packed into Blister packs in Blister Packing Machine, where the Tablets filled in the PVC/PVDC cavity and shall be sealed by Lidding Printed Aluminium Foil.
Bulk Packing
30 Tablets or 60 Tablets shall be bulk packed into HDPE containers, capped and sealed with aluminium tagger wad. The containers shall be labelled with Printed Label of the particular product and Shrink wrapped.
Secondary packing
The Blister packs shall be either packed into Monocartons, each containing one Blister pack or Outer cartons each containing 10 Blister packs.
Such Outer cartons/Packed HDPE containers Shall be Packed into Corrugated Box for ease of transportation.
Storage of finished product
Finished Product after Packaging shall be stacked over Pallets, separately Batchwise in Finished Product Storage area under controlled environmental conditions, until the completion of Testing and Release.
Dispatch
The Finished Product shall be dispatched to Customer Warehouse after the Release from Quality control.